Because the volume of each of the eight corner atoms is shared between eight adjacent cells, each BCC cell contains the equivalent volume of two atoms (one central and one on the corner).Įach corner atom touches the center atom. The primitive unit cell for the body-centered cubic crystal structure contains several fractions taken from nine atoms (if the particles in the crystal are atoms): one on each corner of the cube and one atom in the center. Knowing this and the formula for the volume of a sphere, it becomes possible to calculate the APF as follows: Body-centered cubic Using geometry, and the side length, a can be related to r as: A line can be drawn from the top corner of a cube diagonally to the bottom corner on the same side of the cube, which is equal to 4 r. Face-centered cubicįor a face-centered cubic unit cell, the number of atoms is four. The side of the unit cell is of length 2 r, where r is the radius of the atom. Simple cubicįor a simple cubic packing, the number of atoms per unit cell is one. 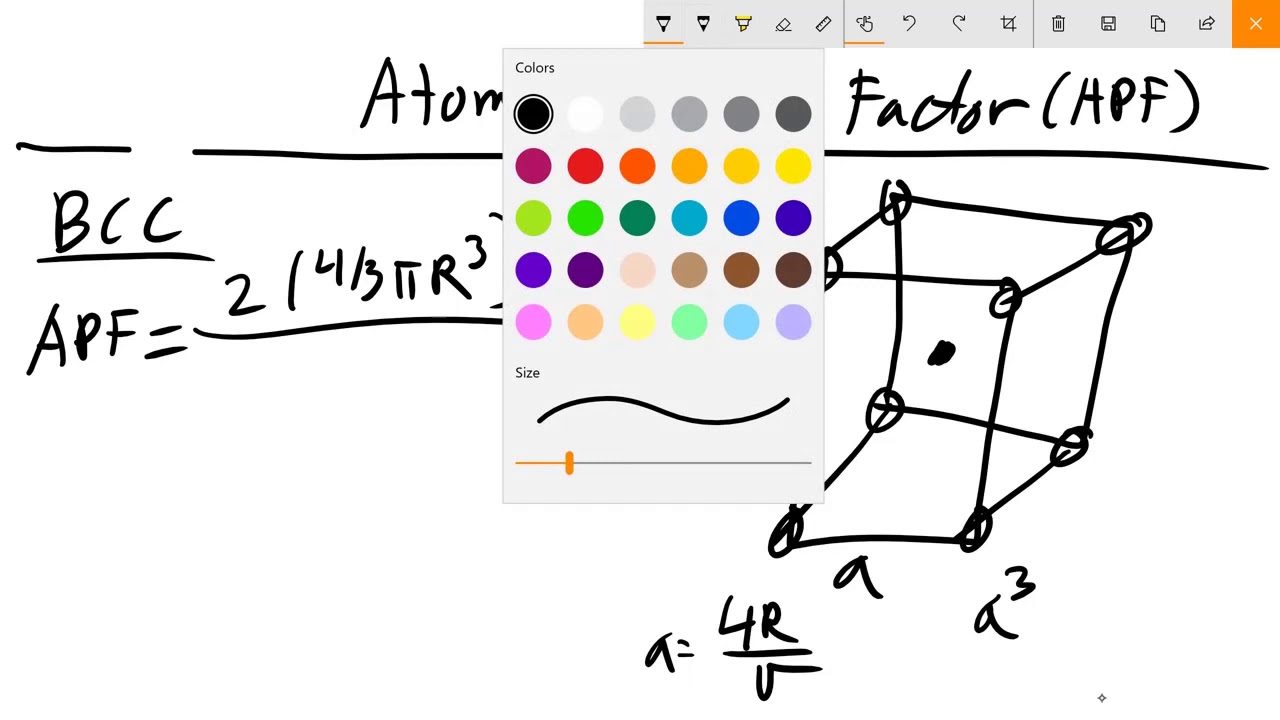
Packing factor or Atomic Packing factor (APF) can be defined as the ratio between the volume of the basic atoms of the unit cell (which represents the volume of all atoms in one unit cell ) to the volume of the unit cell itself.įor cubic crystals, it depends on the radius of atoms and the characterization of chemical bondings.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
